Research Article
Evaluation of the antibacterial sensitivity properties of mannitol salt agar
Evaluación de las propiedades de sensibilidad antibacteriana en agar sal manitol
Falah Hasan Obayes AL-Khikani1* https://orcid.org/0000-0002-8890-7090
Mustafa Sahib Abd-alhussein2 https://orcid.org/0009-0007-9780-5460
Karar Kadhim Madloul2 https://orcid.org/0009-0004-4540-2801
1The Islamic University. College of Medical Technology. Department of Medical Laboratory Technology. Najaf, Iraq.
2Babylon Health Directorate. Al- Hashmia General Hospital. Department of Microbiology. Babylon, Iraq.
*Author for correspondence. Email: falahgh38@gmail.com
ABSTRACT
Background: Staphylococcus spp. are major pathogens causing diverse clinical infections. Evaluating their phenotypic characteristics and antimicrobial response across different culture media is essential for accurate diagnosis.
Objective: To assess vancomycin susceptibility testing performance of Mannitol Salt Agar (MSA) compared with Mueller–Hinton Agar (MHA) against Staphylococcus spp.
Methods: Thirty clinical specimens (urine, wound, ear swabs) were collected. Isolates were identified using standard microbiological methods including MSA for colony color, blood agar for hemolysis, and VITEK-2 confirmation. Vancomycin susceptibility was evaluated on both media. Statistical analysis used t-test, chi-square, and Pearson correlation (p < 0.05).
Results: Staphylococcus spp. were predominantly isolated from urine (60%; p = 0.007). On MSA, 40% showed yellow colonies and 60% pinkish-white (p = 0.27). Hemolysis patterns were 46.7% beta and 53.3% alpha/gamma (p > 0.05). Vancomycin produced significantly larger inhibition zones on MSA (25.3 ± 6.1 mm) than MHA (17.5 ± 5 mm; p < 0.0001). Strong correlation existed between colony color and hemolysis pattern (r = 0.873, p < 0.001).
Conclusions: Vancomycin showed significantly larger inhibition zones on MSA compared to MHA, indicating that high salt content and mannitol alter antibiotic diffusion and/or bacterial phenotypic expression. Larger zones on MSA represent greater apparent susceptibility rather than enhanced resistance detection. Therefore, MSA cannot replace MHA for standardized antimicrobial susceptibility testing, as altered medium composition leads to misleading susceptibility profiles.
Keywords: antimicrobial susceptibility testing; disk diffusion; high-salt medium; mannitol salt agar; Mueller-Hinton agar; Staphylococcus aureus; vancomycin.
RESUMEN
Antecedentes: Staphylococcus spp. son patógenos importantes que causan diversas infecciones clínicas. Evaluar sus características fenotípicas y respuesta antimicrobiana en diferentes medios es esencial para el diagnóstico preciso.
Objetivo: Evaluar el desempeño del Agar Sal Manitol (MSA) comparado con Agar Mueller–Hinton (MHA) para pruebas de susceptibilidad a vancomicina contra Staphylococcus spp.
Métodos: Se recolectaron 30 muestras clínicas (orina, heridas, oído). Los aislamientos se identificaron mediante métodos microbiológicos estándar: MSA para color de colonias, agar sangre para hemólisis y confirmación con VITEK-2. La susceptibilidad a vancomicina se evaluó en ambos medios. Se utilizaron prueba t, ji cuadrado y correlación de Pearson (p < 0.05).
Resultados: Staphylococcus spp. se aisló principalmente de orina (60 %; p= 0,007). En MSA, 40 % de colonias fueron amarillas y 60 % rosadas blanquecinas (p= 0,27). Los patrones de hemólisis fueron 46,7 % beta y 53,3 % alfa/gamma (p> 0,05). La vancomicina produjo zonas de inhibición significativamente mayores en MSA (25,3 ± 6,1 mm) que en MHA (17,5 ± 5 mm; p< 0,0001). Se observó fuerte correlación entre color colonial y hemólisis (r= 0,873; p< 0,001).
Conclusiones: La vancomicina mostró zonas de inhibición significativamente mayores en MSA que en MHA, indicando que el alto contenido de sal y manitol alteran la difusión del antibiótico o la expresión fenotípica bacteriana. Las zonas mayores en MSA representan mayor susceptibilidad aparente y no mejor detección de resistencia. Por tanto, el MSA no puede sustituir al MHA en pruebas estandarizadas de susceptibilidad antimicrobiana.
Palabras clave: agar Mueller-Hinton; agar sal manitol; difusión en disco; medio de alta salinidad; pruebas de susceptibilidad antimicrobiana; Staphylococcus spp.; vancomicina.
Received: 30/12/2025
Approved: 13/04/2026
INTRODUCTION
Staphylococcus is a ubiquitous Gram-positive pathogen renowned for its ability to cause a wide spectrum of infections, from superficial skin and soft tissue infections to invasive diseases such as pneumonia, endocarditis, osteomyelitis, and sepsis.(1) It is one of the leading causes of nosocomial and community-acquired infections globally, with increasing relevance due to its evolving resistance to multiple antibiotics. This organism exhibits a remarkable ability to adapt to various environmental pressures, survive within host immune defenses, and develop persistent infections, making it a critical public health concern.(2)
Although S. aureus is not the most common uropathogen, it is increasingly implicated in urinary tract infections (UTIs), particularly among catheterized patients, the elderly, and individuals with diabetes or immunocompromised status.(3) Its ability to adhere to uroepithelial cells and form biofilms enhances its pathogenicity in the urinary system. In wound infections, S. aureus is more frequently encountered, especially in post-surgical sites and trauma-related injuries. The organism's production of cytotoxins, surface adhesins, and enzymes enables tissue invasion, immune evasion, and chronic colonization.(4)
Blood agar and Mannitol Salt Agar (MSA) are widely used to phenotypically identify S. aureus. Hemolysis, particularly beta-hemolysis, is a hallmark of virulent S. aureus strains and is mediated by cytotoxins like alpha-hemolysin, which causes complete lysis of red blood cells.(5) On MSA, S. aureus ferments mannitol, leading to a color shift from red to yellow, aiding in its differentiation from other coagulase-negative staphylococci. These phenotypic characteristics correlate with strain virulence and clinical outcomes.(6)
The adaptability of S. aureus is further underscored by its resistance development. Methicillin-resistant Staphylococcus aureus (MRSA) is now endemic in many healthcare systems and increasingly identified in community settings. MRSA strains carry the mecA gene, encoding penicillin-binding protein 2a (PBP2a), which has reduced affinity for β-lactam antibiotics.(7) These strains have shown resistance not only to methicillin but also to macrolides, aminoglycosides, and fluoroquinolones, complicating treatment strategies.(8)
For decades, vancomycin has been the mainstay for treating MRSA infections. However, reduced susceptibility has emerged in the form of vancomycin-intermediate S. aureus (VISA) and, more recently, vancomycin-resistant S. aureus (VRSA). VISA is typically associated with thickened cell walls and stepwise mutations in cell wall biosynthesis pathways,(9) whereas VRSA strains acquire the vanA gene from enterococci, leading to altered peptidoglycan precursors that prevent vancomycin binding.(10) These strains have been implicated in therapeutic failures and increased patient mortality.(11)
Staphylococcus aureus has a unique capacity to form biofilms on indwelling medical devices and mucosal surfaces, which significantly enhances its resistance to antibiotics and immune-mediated killing. Biofilms consist of bacterial cells encased in an extracellular polymeric matrix, which limits antibiotic penetration and facilitates persistent infection.(12) Additionally, S. aureus can survive intracellularly in non-professional phagocytes, contributing to recurrent infections despite treatment.(13)
The global epidemiology of S. aureus, particularly MRSA, has evolved dramatically. Molecular typing has revealed several epidemic clones circulating worldwide, including USA300, EMRSA-15, and ST239. These clones are associated with distinct virulence profiles and resistance patterns, emphasizing the need for continuous surveillance and genomic monitoring.(14) Both hospital-acquired (HA-MRSA) and community-acquired (CA-MRSA) strains contribute significantly to the burden of disease, although their transmission dynamics differ.(15)
Given the limitations of current antibiotics, alternative treatment approaches are under investigation. These include newer glycopeptides (dalbavancin, oritavancin), lipoglycopeptides, anti-virulence therapies, and phage therapy. Meanwhile, standardization of antimicrobial susceptibility testing, including media used (e.g., MSA vs. Muller Hinton Agar), remains critical for interpreting inhibition zones accurately.(16) Media composition can influence diffusion rates and bioavailability, thus impacting the reliability of susceptibility profiles.
This study aim to evaluate the feasibility of using Mannitol Salt Agar as an alternative to Mueller-Hinton Agar in measuring the antibiotic susceptibility of Staphylococcus spp. isolates to determine if there are statistically significant differences in the results of antibiotic susceptibility testing when using the two media.
METHODS
Design
This research followed a cross-sectional, descriptive laboratory design. The study focused on comparing the antibacterial activity of vancomycin on Mannitol Salt Agar (MSA) and Mueller-Hinton Agar (MHA) to evaluate possible medium-dependent variations in antibiotic response.
Subjects
The study population consisted of patients attending healthcare facilities in the southern region of Babylon, Iraq, who presented with clinical signs suggestive of bacterial infection. A total of 30 clinical samples were collected between March 2025 and June 2025. These samples included urine, wound swabs, and ear swabs selected through purposive sampling based on clinical suspicion of infection. For each participant, demographic information such as age and sex was recorded to support further correlation analyses.
Variables
The study involved defined microbiological and clinical variables necessary to achieve the objectives. Sample type, colony color on MSA, and hemolysis pattern on blood agar were treated as descriptive and comparative variables related to bacterial characterization. The primary outcome variables were the diameters of vancomycin inhibition zones measured on both MSA and MHA. Additional descriptive variables, including age, sex, and bacterial species identification, were included to assess their potential associations with phenotypic characteristics and antimicrobial susceptibility outcomes.
Procedures
Susceptibility testing was performed using the Kirby–Bauer disk diffusion method following CLSI M100, 34th Edition (2024) guidelines. Vancomycin (30 µg) disks (Oxoid, UK; Lot No. 2384712) were applied to both MSA and MHA plates using a disk dispenser. Quality control was ensured by testing ATCC reference strain Staphylococcus aureus ATCC 25923 in parallel with each batch, with acceptance zones for vancomycin (30 µg) on MHA ranging 17–21 mm per CLSI criteria. MSA (HiMedia Laboratories, Mumbai, India; Lot No. MSA-2025-03) and MHA (Oxoid, UK; Lot No. MHA-2025-02) were prepared according to manufacturer instructions with a standardized agar depth of 4 mm measured at 10 random points per plate. Bacterial suspensions were standardized to 0.5 McFarland turbidity (approximately 1.5 × 10⁸ CFU/mL) using a DensiCHEK densitometer (bioMérieux, France), verified by spectrophotometry at 625 nm (absorbance 0.08–0.10), and inoculated using cotton swabs streaked across the entire agar surface in three directions. Plates were incubated aerobically at 35 ± 2 °C for 16–18 hours in ambient air, with zone diameters measured to the nearest millimeter using a digital caliper (Mitutoyo, Japan) from the back of the plate against a dark background illuminated with reflected light.
Processing
Continuous variables (age, inhibition zone diameters) were summarized as mean ± standard deviation (SD), while categorical variables (colony color, hemolysis pattern, sample type) were expressed as frequencies and percentages. Comparison of inhibition zones between MSA and MHA was performed using the paired samples t-test since the same bacterial isolates were tested on both media, with normality confirmed by the Shapiro-Wilk test and effect size calculated using Cohen's d. Sex differences were analyzed using the independent samples t-test for age and inhibition zones. Associations among categorical variables were assessed using the Chi-square test or Fisher's exact test when expected cell counts were less than 5. Spearman's rank correlation coefficient (ρ) was used to evaluate relationships between categorical variables (colony color, hemolysis type, sample type), while point-biserial correlation was applied for dichotomous variables (sex). The Shapiro-Wilk test was used to assess normality of continuous variables, with non-parametric alternatives (Wilcoxon signed-rank test for paired comparisons; Mann-Whitney U test for independent comparisons) to be applied if normality assumptions were violated. All statistical analyses were performed using SPSS version 26 (IBM, USA) with a significance threshold of p < 0.05.
Bioethical Issues
The study adhered to all applicable institutional and national ethical guidelines. Ethical approval was obtained from the Institutional Ethics Committee under approval code IEC/2025/053, dated March 1, 2025. All clinical samples were handled confidentially, and no personal identifiers were included at any stage of the research. The study involved human-derived samples only, with minimal risk to participants and full compliance with bioethical standards for research involving human subjects.
RESULTS
From a total of 30 isolates (S. aureus), females were more distributed 20 (66.7%) compared to males 10 (33.3%) without significant difference (p = 0.06).
The prevalence of staphylococcus spp. in the urine samples was high 18 (60%), lower than wound 7 (23.3%) and ear swab 5 (16.7%) with high significant differences (p = 0.007).
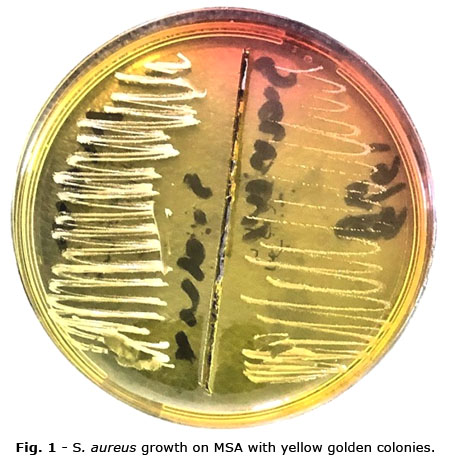
Color of colonies on MSA showed that yellow color was found in 12 (40%) samples while pinkish white color was observed in 18 (60%) of samples without significant differences between them (p= 0.27) (Fig. 1; Fig. 2).
Beta hemolysis observed in 14 (46.7%) samples on blood agar while alpha or gamma hemolysis was detected in 16 (53.3%) Without significant difference (p > 0.05).
Mean age of males was 37.60 ± 18.41 years, while the mean age of females was 32.05 ± 11.56 years. The difference was not statistically significant (p = 0.31).
For Vancomycin- Mueller H agar, the mean value for males was 18.90 ± 3.72, compared to 16.90 ± 5.55 for females, with no statistically significant difference (p = 0.31).
Regarding Vancomycin- Mannitol S agar, males had a mean value of 26.30 ± 4.96, while females had 24.85 ± 6.66, and the difference was not statistically significant (P = 0.54) (table 1).
Paired comparison of vancomycin inhibition zones showed significantly larger diameters on Mannitol Salt Agar (25.3 ± 6.1 mm) compared to Mueller-Hinton Agar (17.5 ± 5 mm; paired t-test, p < 0.0001, Cohen's d = 1.45) (table 2).
The correlation analysis between different variables revealed the following findings (table 3). Sex showed a negative correlation with colony color (r = -0.196, p = 0.300), no correlation with type of hemolysis (r = 0.000, p = 1.000), and a weak correlation with sample (r = 0.047, p = 0.804). None of these correlations were statistically significant.
Sample had weak correlations with colony color (r = 0.090, p = 0.637) and type of hemolysis (r = 0.084, p = 0.660), both of which were not statistically significant.
Colony color showed a strong and statistically significant positive correlation with type of hemolysis (r = 0.873, p = 0.000), indicating a strong association between these two variables.
DISCUSSION
Among the 30 Staphylococcus aureus isolates examined, the inhibition zone of vancomycin in Muller Hinton agar was 17.5± 5 mm. This high susceptibility rate is encouraging, particularly in an era of rising multidrug resistance. Vancomycin remains one of the most effective antibiotics for serious infections caused by Gram-positive cocci, especially methicillin-resistant Staphylococcus aureus (MRSA).)18) However, resistance is gradually emerging in clinical settings, especially in high-risk patient populations such as those undergoing dialysis or receiving long-term care.)19) This highlights the necessity of routine antimicrobial susceptibility testing prior to empirical treatment, as resistance could compromise the clinical utility of vancomycin.(20)
Out of the 30 samples, females represented a greater proportion (66.7%) compared to males (33.4%), although this difference was not statistically significant (p = 0.06). This observation is consistent with established epidemiology, where females are more frequently affected by S. aureus urinary tract infections (UTIs) due to their shorter urethral anatomy, hormonal influences, and frequent healthcare contact.(21) Still, the insignificant P-value suggests that gender was not a determining factor for the distribution of S. aureus infections in this specific population, emphasizing the multifactorial nature of infection susceptibility.
The highest frequency of S. aureus was observed in urine samples (60%), followed by wound samples (23.3%) and ear swabs (16.7%), with statistically significant differences between these categories (p = 0.007). This pattern reflects the adaptability of S. aureus to colonize both mucosal and cutaneous environments. Its increasing presence in urine is particularly notable, possibly facilitated by catheterization, biofilm formation on uroepithelial cells, and co-morbidities such as diabetes.(22) Studies have also shown that urinary S. aureus may result in hematogenous spread in bacteremic patients, underlining its clinical significance.(23)
On MSA, 12 isolates (40%) formed yellow colonies indicative of mannitol fermentation—characteristic of S. aureus—while 18 isolates (60%) produced pinkish-white colonies, suggesting non-fermenting staphylococci or other species. Although this difference was not statistically significant (p = 0.27), the presence of yellow colonies on MSA serves as a classic phenotypic confirmation of S. aureus. The ability to ferment mannitol is regulated by metabolic genes that can vary with strain virulence and environmental stress.(24)
Beta hemolysis was observed in 14 (46.7%) of the isolates, whereas 16 (53.3%) exhibited alpha or gamma hemolysis. No statistically significant difference was observed (p > 0.05). Hemolysins, particularly alpha- and beta-toxins, are virulence factors that enable S. aureus to lyse red blood cells and evade immune responses.(25) Their variable expression may reflect differing levels of virulence across strains. Hemolytic profiles can also inform potential tissue damage and severity in systemic infections.
Comparison between males and females for age, and for vancomycin inhibition zones on Muller Hinton and Mannitol Salt Agars, revealed no significant differences. This implies that host sex does not play a prominent role in determining the inhibition efficiency of vancomycin or the phenotypic behavior of S. aureus. This finding supports previous observations that S. aureus virulence and antibiotic resistance are primarily driven by bacterial genetics and environmental exposure, rather than host gender.(26)
A significant difference was observed between the inhibition zones of vancomycin on Mueller-Hinton Agar (17.5 ± 5 mm) versus Mannitol Salt Agar (25.3 ± 6.1 mm), with a P-value < 0.0001. Importantly, larger inhibition zones on MSA indicate greater apparent susceptibility due to altered antibiotic diffusion in the high-salt medium, not enhanced resistance detection. This medium-dependent variation underscores why MSA cannot substitute MHA for standardized antimicrobial susceptibility testing, as it would yield misleading susceptibility profiles.(27)
The strongest correlation was identified between colony color and hemolysis type (r = 0.873, p < 0.001), indicating that isolates fermenting mannitol (yellow colonies) were more likely to show beta-hemolysis, a virulence marker of S. aureus. This suggests that fermentation capacity and hemolytic ability may be co-regulated or reflect the metabolic vigor of certain strains.(28) Other correlations, including sex vs. colony traits or sample type vs. hemolysis, were weak and statistically non-significant, reflecting that environmental or procedural variables may have greater influence than demographic factors.
Vancomycin produced significantly larger inhibition zones on Mannitol Salt Agar (25.3 ± 6.1 mm) compared to Mueller-Hinton Agar (17.5 ± 5 mm; p < 0.0001). These larger zones indicate greater apparent susceptibility due to altered antibiotic diffusion and phenotypic expression in the high-salt medium, rather than enhanced resistance detection. Therefore, MSA cannot be used as a substitute for MHA in standardized antimicrobial susceptibility testing, as the medium-dependent variation leads to misleading susceptibility profiles that could compromise clinical interpretation.
BIBLIOGRAPHIC REFERENCES
1. Gherardi G. Staphylococcus aureus Infection: Pathogenesis and Antimicrobial Resistance [Internet]. Int J Mol Sci. 2023;24(9):8182. DOI: 10.3390/ijms24098182
2. Al-Mebairik NF, El-kersh TA, Al-Sheikh YA, Marie MA. A review of virulence factors, pathogenesis, and antibiotic resistance in Staphylococcus aureus [Internet]. Rev Med Microbiol. 2016;27(2):51-56. DOI: 10.1097/MRM.0000000000000067
3. Nogueira JO, da Silva JD, de Pascoa Junior JG, Pereira TC, Bezerra CC. Staphylococcus aureus resistente em unidade de terapia intensiva [Internet]. Res Soc Dev. 2023;12(6):e7312. DOI: 10.33448/rsd-v12i6.42006
4. Nikolic P, Mudgil P. The Cell Wall, Cell Membrane and Virulence Factors of Staphylococcus aureus and Their Role in Antibiotic Resistance [Internet]. Microorganisms. 2023;11(2):259. DOI: 10.3390/microorganisms11020259
5. Kumar S, Singh S, Dhanjal DS. Pathogenesis and Antibiotic Resistance of Staphylococcus aureus. In: Arora G, Sajid A, Kalia VC, editors. Microbial Pathogenesis: Strategies for Diagnosis and Therapy [Internet]. Singapore: Springer; 2020. p. 1-20. DOI: 10.1007/978-981-15-1695-5_7
6. Mukherjee R, Priyadarshini A, Pandey RP, Raj VS. Antimicrobial Resistance in Staphylococcus aureus [Internet]. In: Islam Aqib A, Rodríguez-Morales AJ. Insights Into Drug Resistance in Staphylococcus aureus. London: IntechOpen; 2021. DOI: 10.5772/intechopen.96888
7. Monaco M, Araujo FP, Cruciani M, Coccia EM, Pantosti A. Worldwide Epidemiology and Antibiotic Resistance of Staphylococcus aureus [Internet]. Curr Top Microbiol Immunol. 2016;409:21-56. DOI: 10.1007/82_2016_3
8. Stefani S, Goglio A. Methicillin-resistant Staphylococcus aureus: related infections and antibiotic resistance [Internet]. Int J Infect Dis. 2010; (Suppl 4):S19-22. DOI: 10.1016/j.ijid.2010.05.003
9. Hu Q, Peng H, Rao X. Molecular events for promotion of vancomycin resistance in vancomycin intermediate Staphylococcus aureus [Internet]. Front Microbiol. 2016;7:1601. DOI: 10.3389/fmicb.2016.01601
10. Selim S, Faried OA, Almuhayawi MS. Incidence of Vancomycin-Resistant Staphylococcus aureus Strains among Patients with Urinary Tract Infections [Internet]. Antibiotics. 2022;11(3):408. DOI: 10.3390/antibiotics11030408
11. Patel H, Rawat S. A genetic regulatory see-saw of biofilm and virulence in MRSA pathogenesis [Internet]. Front Microbiol. 2023;14:1204428. DOI: 10.3389/fmicb.2023.1204428
12. Tasneem U, Majid M, Mehmood K. Co-occurrence of antibiotic resistance and virulence genes in Methicillin Resistant Staphylococcus aureus (MRSA) isolates from Pakistan [Internet]. Afr Health Sci. 2022;22(2):567-77. DOI: 10.4314/ahs.v22i1.57
13. Mesrati I, Saidani M, Jemili M. Virulence determinants, biofilm production and antimicrobial susceptibility in Staphylococcus aureus causing device-associated infections [Internet]. Int J Antimicrob Agents. 2018;52(3):322-7. DOI: 10.1016/j.ijantimicag.2018.05.004
14. Alkuraythi DM. Virulence Factors and Pathogenicity of Staphylococcus aureus [Internet]. In: Bustos J, Valdés JJ, Hamdan A, Rodríguez AJ. Advances and Perspectives of Infections Caused by Staphylococcus aureus. London: IntechOpen; 2024. DOI: 10.5772/intechopen.1006633
15. Graf AC, Leonard A, Schauble M. Virulence Factors Produced by Staphylococcus aureus Biofilms Have a Moonlighting Function Contributing to Biofilm Integrity [Internet]. Mol Cell Proteomics. 2019;18(5):1036-52. DOI: 10.1074/mcp.RA118.001120
16. Ballah FM, Islam MS, Rana M. Virulence Determinants and Methicillin Resistance in Biofilm-Forming Staphylococcus aureus from Food Sources [Internet]. Antibiotics. 2022;11(11):1666. DOI: 10.3390/antibiotics11111666
17. Humphries R, Bobenchik AM, Hindler JA, Schuetz AN. Overview of changes to the CLSI performance standards for antimicrobial susceptibility testing, M100 [Internet]. J Clin Microbiol. 2021;59(3):e00213-21. DOI: 10.1128/JCM.00213-21
18. Yousefi M, Fallah F, Arshadi M, Pourmand MR, Hashemi A, Pourmand G. Identification of tigecycline- and vancomycin-resistant Staphylococcus aureus strains in urinary tract infection patients [Internet]. New Microbes New Infect. 2017;19:8-12. DOI: 10.1016/j.nmni.2017.05.009
19. McGuinness WA, Malachowa N, DeLeo FR. Vancomycin Resistance in Staphylococcus aureus [Internet]. Yale J Biol Med. 2017 [access: 13/04/2026];90(2):269-281. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC5482303/
20. Tong SYC, Davis JS, Eichenberger E, Holland TL, Fowler VG. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management [Internet]. Clin Microbiol Rev. 2023;36(3):e00013-23. DOI: 10.1128/cmr.00134-14
21. Cernohorska L. Antibiotic resistance and biofilm formation in Staphylococcus aureus strains isolated from urine [Internet]. Klin Mikrobiol Infekc Lek. 2010 [access: 13/04/2026];16(5):196-198. Available from: https://pubmed.ncbi.nlm.nih.gov/21243597/
22. Wang X, Zhan P, Zhang Q, Li R, Fan H. Staphylococcus aureus acquires resistance to vancomycin via CXCL10 [Internet]. Int Immunopharmacol. 2024;132:111780. DOI: 10.1016/j.intimp.2024.111780
23. Sahle B, Merid Y. Prevalence and antibiotic resistance of Staphylococcus aureus in wound infections [Internet]. J Infect Dev Ctries. 2024;18(10):1530-8. DOI: 10.3855/jidc.19023
24. Stojanovic-Radic Z, Dimitrijevic M, Stankovic N, Aleksic A, Pejcic M. Frequency of isolation and antibiotic resistance patterns of bacterial isolates from wound infections [Internet]. Biol Nyssana. 2016;7:151-158. DOI: 10.5281/zenodo.200414
25. Vandenesch F, Lina G, Henry T. Staphylococcus aureus hemolysins, bi-component leukocidins, and cytolytic peptides: A redundant arsenal of membrane-damaging virulence factors? [Internet]. Front Cell Infect Microbiol. 2012;2:12. DOI: 10.3389/fcimb.2012.00012 Available from: https://www.frontiersin.org/articles/10.3389/fcimb.2012.00012/full
26. de Albuquerque VV, Flores VC, Zeron RM, Godoi BB, Eulalio Filho WM, dos Santos BA. Study of vancomycin resistance among Staphylococcus aureus [Internet]. Amadeus Int Multidiscip J. 2019;3(6):31-37. DOI: 10.14295/aimj.v3i6.58
27. Olayinka BO, Olayinka AT, Onaolapo JA, Olurinola PF. Patterns of resistance to vancomycin and other agents in staphylococcal isolates [Internet]. Afr J Clin Exp Microbiol. 2005;6(1):21-27. DOI: 10.4314/ajcem.v6i1.7395
28. Pal M, Kerorsa GB, Marami LM, Kandi V. Epidemiology, pathogenicity, animal infections, antibiotic resistance, public health significance and economic impact of Staphylococcus aureus: a review [Internet]. Am J Public Health Res. 2020;8(1):14-21. DOI: 10.12691/ajphr-8-1-3
Conflicts of interest
None of conflicts of interest in relation to the work.
Authorship contribution
Conceptualization: Falah Hasan Obayes AL-Khikani, Mustafa Sahib Abd-alhussein, Karar Kadhim Madloul.
Data curation: Falah Hasan Obayes AL-Khikani.
Formal analysis: Falah Hasan Obayes AL-Khikani, Karar Kadhim Madloul.
Research: Falah Hasan Obayes AL-Khikani.
Methodology: Mustafa Sahib Abd-alhussein, Karar Kadhim Madloul.
Writing –original draft: Falah Hasan Obayes AL-Khikani.
Writing –review and editing: Mustafa Sahib Abd-alhussein.
Data availability
Raw data is stored in Excel files and is available from the corresponding author upon reasonable request. Email: falahgh38@gmail.com